Experimental data on the biodegradation of the herbicide glyphosate by the soil fungi trichoderma inhamatum
Scientific articles
Kairat K. Kunanbayev, Galina N. Churkina*, Irina V. Rukavitsina, Dana Yerpasheva
ABSTRACT
Soil fungi of the genus Trichoderma spp. have high agricultural value as an antagonist to phytopathogenic microorganisms. The aim of our studies was to investigate the biodestructive activity of Trichoderma inhamatum with respect to the herbicide containing an active substanceglyphosate under in vitro and in situ conditions. Identification of the strain was carried out by polymerase chain reaction. Activity of the strain was assessed by the rate of radial growth of the colony on the mineral medium M9 with glyphosate and introduction of the strain in the form of an aqueous suspension into the soil, on glyphosate backgrounds. The growth of Tr. inhamatum on a mineral medium with a pesticide was less than on the control. Field tests of the strain - destructor showed changes in the enzymatic activity of soil and biomass of microorganisms. The reduction of residual amounts of pesticides in the M9 mineral media and soil indicate that Tr. Inhamatum is capable of destroying glyphosate.
KEYWORDS: pesticides destruction, glyphosate, Trichoderma inhamatum, enzymatic activity.
INTRODUCTION Glyphosate (N-(phosphonomethyl)-glycine, C3H8NO5P) is a nonselective systemic herbicide used to control weeds, especially perennials. Glyphosate occupies one of the leading places in the world by production and usage [1]. The action of glyphosate is based on the inhibition of 5-enolpyruvil shikimate3-phosphate synthase, which leads to the depletion of the basic aromatic amino acids vital for plants. In agriculture, glyphosate is intensively used in zero tillage. The long-term application of glyphosate leads to certain shifts in the soil microbiota. Studies conducted by Monsanto show a high degradation activity of the herbicide with a decay period of 20 days [2]. Nevertheless, the prolonged usage of herbicide for 30 years leads to gradual accumulation of glyphosate in the soil. The level of pollution depends on the soil - climatic conditions of the soil, tillage methods, climatic conditions and other factors. A considerable number of works have been devoted to the investigations on the interaction of soil microorganisms with pesticides. There are also many studies on the biodegradation of herbicides by fungi [3-5]. Most research on the degradation of glyphosate is aimed at the isolation of bacteria-destructors and the study of their metabolic pathways [6, 7].There are few studies on biodegradation of glyphosate with a Trichoderma fungus. However, this fungi genus is of some interest because they are natural antagonists of the phytopathogenic microflora and are partly involved in the destruction of cellulose [8, 9]. In this regard, we believe that studies of glyphosates biodegradation with fungi of the genus Trichoderma are very relevant, since they will solve environmental problems of soil contamination with pollutants.
MATERIAL AND METHODS
In our studies, the growth ability of Trichoderma was studied in vitro on the media containing glyphosate at various concentrations. In the field, dynamics of the microbial biomass and enzymatic activity were identified when a culture fluid of the Trichoderma fungus was introduced into the soil treated with glyphosate.
RESEARCH RESULTS
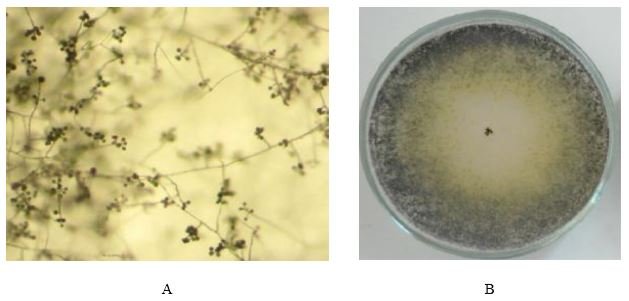
The colony of Tr. inhamatum (strain F6-2014) fungus at initial stage of growth on Czapek’s-Dox medium was colorless, then acquired a green color and occupied almost the entire area of the Petri dish. Microscopy showed hyphae 1.5-8.0 μm in diameter. Thin hyphae were colorless, strongly branched, septate, while thick hyphae are slightly branched, septate, formed swellings and outgrowths. Conidiophores in the form of short branches of the mycelium were poorly visible. Conidia are unicellular, globular or ellipsoidal 2.3-3.0 × 2.0-2.6 μm in size, light or colorless, often crowded into small heads (Figure 1).
 Figure 1 – Fungus colony of Tr. inhamatum on the Czapek’s-Dox medium (А) and conidia formation on 15th day of cultivation (В,) (150х)
Figure 1 – Fungus colony of Tr. inhamatum on the Czapek’s-Dox medium (А) and conidia formation on 15th day of cultivation (В,) (150х)When studying the growth and development of Tr.inhamatum (strain F6-2014) on a medium containing various concentrations of glyphosate, a change in the growth dynamics was established. The first 15 days, the radial growth of colonies with glyphosate (1 mg/l) was 1.75 times smaller than the control (Table 1). The cultivation of the strain on a medium containing glyphosate at a concentration of 10 mg/l led to 1.50 times decrease in the growth rate of the colony. Thus, after 15 days of cultivation, there was a statistically significant difference between the control and variants with different concentrations of the herbicide. At the same time, during this incubation period, there were no significant differences observed in the diameter of the colony between two variants: (1 mg/L) and (10 mg/L). On the thirtieth day of cultivation of Tr.inhamatum fungus on the medium with glyphosate (1 mg/l), the colony diameter increased by only 1 mm, at a concentration of 10 mg/L by 5 mm. In comparison to the control, colonies diameters on a medium with glyphosate concentrations at 1 mg/L and 10 mg/L were 1.89 and 1.48 times smaller than the controls, respectively. A statistically significant difference was identified between two variants 1.0 mg/L and 10.0 mg/L and control. At the same time, a significant increase in the colony growth from day 15 to day 30 was recorded only in a medium containing glyphosate at a concentration of 10 mg/l. On the 45th day of cultivation on a medium with 1 mg/l glyphosate, the colony growth nearly ceased. Difference in the growth rate between control and the variant (1 mg/l) was 2.48 fold. The radial growth of the colony on a medium with a concentration of 10 mg/l was 41 mm that is 31 mm smaller than the control. A statistically significant difference in growth rate between all variants was recorded.
Table 1 – The effect of glyphosate on the radial growth rate of Tr.inhamatum colony and pH
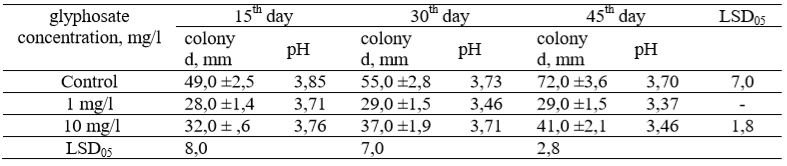 Note: D is the diameter of the colony
Note: D is the diameter of the colonyChange in the pH of the medium was noted in all variants, including control. The control pH value of the mineral medium became more acidic on 0.12 units after 30 days and on 0.15 units after 45 days, with the initial pH value of from 3.85. On the variant (1 mg/l) with pH 3.71, the shift to more acidic medium was sharper on 0.25 units after 30 days and 0.34 units on day change in the pH from 3.76 on the day15, to 3.46 on the day45. A more acidic environment on variants with glyphosate should be noted at the first moment of fixing the pH of the medium on 15th day. Over a period of 100 days there was a change in the biomass dynamics of microorganisms in field experiments. At the first day, the smallest biomass of microorganisms was on the control (Table 2). In the same period, variants with the application of the herbicide (glyphosate active ingredient) with the consumption rate of (2.0-4.0 l/ha) increased microbial biomass 1.70 times comparing to the control and by 2.60 times, when the consumption rate was 6.0 l/ha. Statistically significant differences were observed not only between the control and variants with herbicides, but also between the variants with the maximum and minimum application rates of 2.0 and 4.0 l/ha, respectively.
Table 2 – The dynamics of changes in the biomass of microorganisms and urease under the influence of micromycetes Tr.inhamatum, μg/g soil
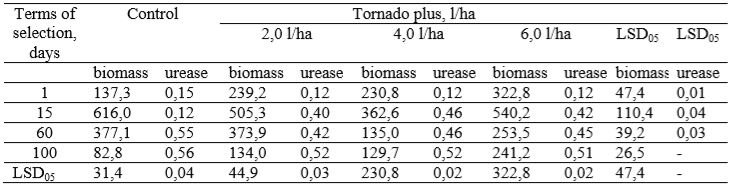 On the 15th day there was an increase in the biomass of microorganisms. On the control variant, it increased 4.46 times and reached the value of 616 mg/kg. The microbial biomass also increased in herbicide variants 2.11 fold for 2.0 l/ha, 1.57 fold for 4.0 l/ha, and 1.67 fold for 6.0 l/ha. Statistically significant differences were recorded between the control and herbicide variants (variants with herbicides application), except for the maximum consumption rate (6.0 l/ha). On the day 60 there was a decrease in the biomass of microorganisms in the control thus becoming 1.63 times smaller. For the herbicide variants the values of microbial biomass decline composed 1.35, 2.68 and 2.13 fold for (2.0 l/ha), (4.0 l/ha) and (6.0 l/ha) respectively. The difference between the control and the herbicide variants was at the rates of glyphosate consumption (4.0 l/ha) and (6.0 l/ha). On the control variant, the microbial biomass was superior to other variants. At day 45, the biomass of microorganisms significantly decreased 4.55 and 2.79 times in both control and the variant with a minimum rate of glyphosate consumption. The urease activity is one of the main indicators characterizing soil biogeneity. On the first day, urease activity on the control was higher than on herbicidal variants (Table 2). On the 15th day there was a slight decrease in urease activity on the control, but it was not statistically reliable. On the 60th day of observations, urease activity increased by 4.58 times in the control. On day 100, the level of enzymatic activity in the control did not change. Also, there were no differences observed between control and glyphosate variants.
On the 15th day there was an increase in the biomass of microorganisms. On the control variant, it increased 4.46 times and reached the value of 616 mg/kg. The microbial biomass also increased in herbicide variants 2.11 fold for 2.0 l/ha, 1.57 fold for 4.0 l/ha, and 1.67 fold for 6.0 l/ha. Statistically significant differences were recorded between the control and herbicide variants (variants with herbicides application), except for the maximum consumption rate (6.0 l/ha). On the day 60 there was a decrease in the biomass of microorganisms in the control thus becoming 1.63 times smaller. For the herbicide variants the values of microbial biomass decline composed 1.35, 2.68 and 2.13 fold for (2.0 l/ha), (4.0 l/ha) and (6.0 l/ha) respectively. The difference between the control and the herbicide variants was at the rates of glyphosate consumption (4.0 l/ha) and (6.0 l/ha). On the control variant, the microbial biomass was superior to other variants. At day 45, the biomass of microorganisms significantly decreased 4.55 and 2.79 times in both control and the variant with a minimum rate of glyphosate consumption. The urease activity is one of the main indicators characterizing soil biogeneity. On the first day, urease activity on the control was higher than on herbicidal variants (Table 2). On the 15th day there was a slight decrease in urease activity on the control, but it was not statistically reliable. On the 60th day of observations, urease activity increased by 4.58 times in the control. On day 100, the level of enzymatic activity in the control did not change. Also, there were no differences observed between control and glyphosate variants.CONCLUSION
1. The Tr. inhamatum F6-2014 fungus strain was identified based on the study of morphological and cultural properties and the method of determining the direct nucleotide sequence of the ITS region (intergenic transcribed region). Tr. inhamatum F6-2014 is able to utilize glyphosate in a dose of 1 mg/l and 10 mg/l. A more acidic reaction of the M9 mineral medium was observed during fungi cultivation with glyphosate.2. The use of Tr.inhamatum F6-2014 liquid culture on variants with different concentrations of glyphosate promoted an increase in microbial biomass in the first day from 1.7 to 2.3 times. The urease activity on variants with glyphosate application exceeded the control. 3. Results of the study showed that the fungus Tr. Inhamatum F6-2014 can be used as a bio destructor in conditions of strong soil contamination with glyphosate.
REFERENCES
[1]Baylis A. D. Why glyphosate is a global herbicide: strengths, weaknesses and prospects // Pest Management Science. ‒ 2000. ‒ T. 56, № 4. ‒ C. 299-308.
[2]Rueppel M. L., Brightwell B. B., Schaefer J., Marvel J. T. METABOLISM AND DEGRADATION OF GLYPHOSATE IN SOIL AND WATER // Journal of Agricultural and Food Chemistry. ‒ 1977. ‒ T. 25, № 3. ‒ C. 517-528.
[3]Diaz J. M. C., Delgado-Moreno L., Nunez R., Nogales R., Romero E. Enhancing pesticide degradation using indigenous microorganisms isolated under high pesticide load in bioremediation systems with vermicomposts // Bioresource Technology. ‒ 2016. ‒ T. 214. ‒ C. 234-241.
[4]Erguven G. O., Bayhan H., Ikizoglu B., Kanat G., Nuhoglu Y. The capacity of some newly bacteria and fungi for biodegradation of herbicide trifluralin under agiated culture media // Cellular and Molecular Biology. ‒ 2016. ‒ T. 62, № 6. ‒ C. 74-79.
[5]Singh B., Singh K. Microbial degradation of herbicides // Critical Reviews in Microbiology. ‒ 2016. ‒ T. 42, № 2. ‒ C. 245-261.
[6]Al-Arfaj A., Abdel-Megeed A., Ali H. M., Al-Shahrani O. Phyto-Microbial Degradation of Glyphosate in Riyadh Area // Journal of Pure and Applied Microbiology. ‒ 2013. ‒ T. 7, № 2. ‒ C. 1351-1365.
[7]Ermakova I. T., Kiseleva N. I., Shushkova T., Zharikov M., Zharikov G. A., Leontievsky A. A. Bioremediation of glyphosate-contaminated soils // Applied Microbiology and Biotechnology. ‒ 2010. ‒ T. 88, № 2. ‒ C. 585-594.
[8]Bouchiat R., Veignie E., Grizard D., Soebert C., Vigier M., Rafin C. Ability of filamentous fungi to degrade four emergent water priority pollutants // Desalination and Water Treatment. ‒ 2016. ‒ T. 57, № 15. ‒ C. 6740-6746.
[9]Arfarita N., Imai T., Kanno A., Yarimizu T., Sun X. F., Wei J., Higuchi T., Akada R. The petential use of Trichoderma viride FRP3 in biodegratdion of the herbicide glyphosate // Biotechnology & Biotechnological Equipment. ‒ 2013. ‒ T. 27, № 1. ‒ C. 3518-3521.
2 273 -рет қаралды
































